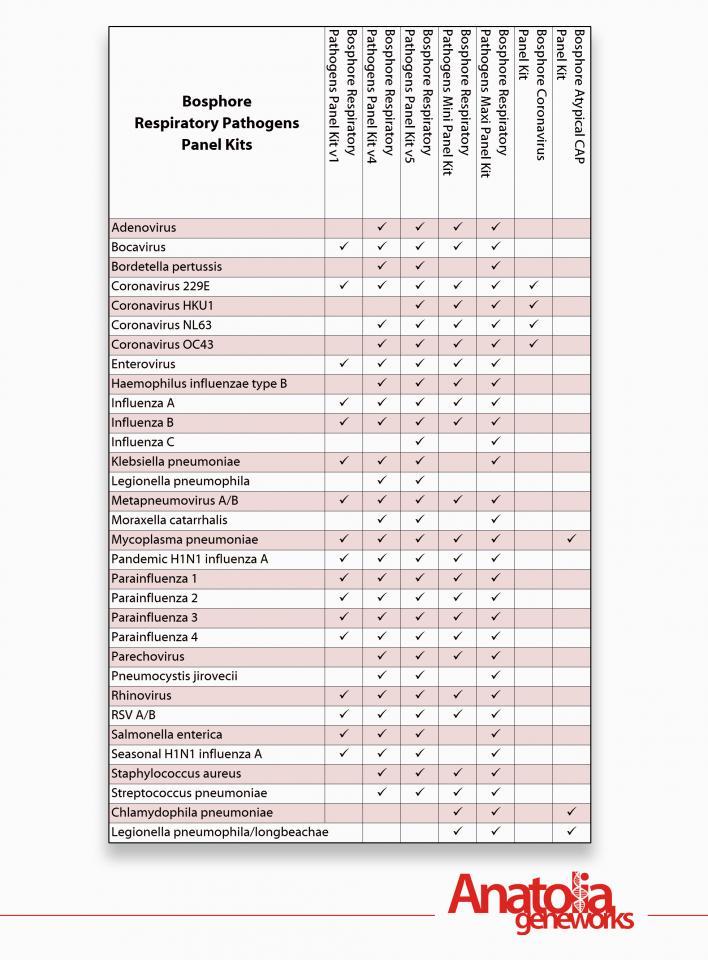
Bosphore Respiratory Pathogens Real-Time PCR Panel Kits
Anatolia Geneworks Panel Kit Solutions for multiplex detection of respiratory pathogens provide high specificity and sensitivity in shorter processing time compared to diagnosis by culture or rapid tests.
Quality Certificate
IVD-CE certified
Method
Taqman principle with dual labelled probes
Packaging
25-50-100 reaction/box packages fit different scales of consumption
Internal Control
Internal control in each tube
Sample Types
Respiratory samples, nasopharyngeal swab specimens, sputum, nasopharyngeal aspirate, bronchoalveolar lavage
Thermal Protocol
All parameters at the same thermal protocol
Platforms
Compatible with different RT PCR instruments
Shelf Life
18 months. Superior to market competitors
Kit Format
Ready to use PCR Master mixes. User friendly
Extraction Kits
Works efficiently with various extraction systems (for IVD purposes)
Channels
FAM, HEX/VIC/JOE, Texas Red/ROX, Cy5